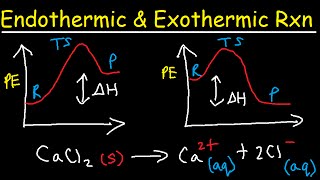
Exergonic vs endergonic reaction diagrams
Вставка
- Опубліковано 3 тра 2022
- ERROR: I keep saying deltaG when I just mean G (Gibbs free energy). The delta is referring to the change. The axis is just G and deltaG is the difference between the reactants and the products.
- Наука та технологія



![ОРБАН приехал к ПУТИНУ и "отчитался" о поездке в Киев 😁 [Пародия]](http://i.ytimg.com/vi/_0ukDovOWq8/mqdefault.jpg)




The smoothest explaining literally. Thank you, and Merry Christmas& Happy new year btw!
Thank you ❤
Thank you for your explanation!