Enthalpy Change of Reaction & Formation - Thermochemistry & Calorimetry Practice Problems
Вставка
- Опубліковано 3 сер 2016
- This chemistry video tutorial focuses on the calculation of the enthalpy of a reaction using standard molar heats of formation, hess law, and calorimetry. This video contains plenty of notes, formulas / equations, examples, and practice problems to help you pass your next exam / general chemistry test on thermochemistry and thermodynamics. Here is a list of topics:
First Law of Thermodynamics:
• First Law of Thermodyn...
Thermochemistry Equations:
• Thermochemistry Equati...
Internal Energy, Heat, and Work:
• Internal Energy, Heat,...
Thermochemical Equations:
• Thermochemical Equations
Specific Vs Molar Heat Capacity:
• What Is The Difference...
________________________________
Basic Calorimetry Problems:
• How To Solve Basic Cal...
Final Temperature Calorimetry Problems:
• Final Temperature Calo...
Latent Heat of Fusion & Vaporization:
• Latent Heat of Fusion ...
Coffee Cup Calorimeter:
• Coffee Cup Calorimeter...
More Calorimeter Problems:
• Bomb Calorimeter vs Co...
__________________________________
Specific Heat Capacity Problems:
• Specific Heat Capacity...
Hess Law Problems:
• Hess's Law Problems & ...
More Hess Law Problems:
• Hess Law Chemistry Pro...
Enthalpy of Formation & Heat Combustion:
• Enthalpy of Formation ...
Enthalpy Practice Problems:
• Enthalpy Change of Rea...
__________________________________
Speed of Light, Frequency, & Wavelength:
• Speed of Light, Freque...
Final Exams and Video Playlists:
www.video-tutor.net/
Full-Length Videos and Worksheets:
/ collections


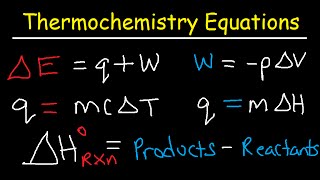






Final Exams and Video Playlists: www.video-tutor.net/
Full-Length Videos & Worksheets: www.patreon.com/MathScienceTutor/collections
girl.. can u like raise your voice omg
You"re a better teacher than most college professors bro... you're the reason why I have an A in chem going into this exam lol
Thanks.
Lmao
The Organic Chemistry Tutor I appreciate for this you helped me a lot
If it’s not rude asking, do you only learn this at college level?
@@axx9149 im having this in my senior year
I seriously hope whatever your heart desires comes to you, you have helped me in ways I can’t describe, you’re a god sent teacher for us hopeless students. Thank you!!
I liked your comment and then unliked it because it was on 100 likes before.😂 i don't want to make it 101.. It will look ugly then😂😂lmao i am such an idiot.
@@saamyarana2096 its 113 now u can like it
@@preetimainali7210 yes😂😂
@@saamyarana2096 😅😂🤣
I know right, we need to reward this man somehow.
as an engineering student, you literally save my life before every exam and i really appreciate that. thank you
@Memati CR harbi
@@castrocuba4937 şaka maka öyle
you are the best, I am completing my Degree this year, your videos have been so much helpful for the past three years . thank you
At 2:42 , the reason it's products-reactants and not the other way around is because it is the enthalpy change for formation, not dissociation. I was confused about this for a moment, hope this helps if someone else has the same issue.
how do you know if its formation or dissolution
You are the closest thing to an angel I've ever known
Thank you so so much 😊 My chemistry teacher had me really confused but you make it seem so easy. Honestly we need more teachers like you
I didnt understand anything
Professor Organic Chemistry Tutor, thank you for an exceptional explanation of Enthalpy Change of Reaction and Formation in AP/General Chemistry. Although the problem selection is excellent, I am confused about some of the problems in this video. Professor Organic Chemistry Tutor, thanks to the viewers for finding and correcting the error(s) in this video.
Your teaching, explanitions, and examples are a tremendous help to myself and so many of my classmates. Our Chem 102 professor just throws terms and formulas at us with zero context and then we have to basically teach ourselves the concepts to be able to use said formulas. I'm sure he's got tenure. Sucks all around.
Thank you so much for this 😭 you are saving me from failing chemistry
im soooo glad i came across this channel i was about to lose hope in passing my general chem course at uni
You deserve an award for this amazing explanation and saving us
I honestly don't know what I would do without you. Thank You
You helped me a lot thanks Mr Organic Chemistry Tutor.
I'm glad I just tune my chem prof out and teach myself online. This isn't too terrible to calculate.
Thank you so much for all your videos especially on chemistry thanks to you I actually understand this
Better than my teacher. Thxs so much for making the effort to make these videos.
You’re so good. The best chemistry teacher ever!!!!!
I've been watching your videos until now I can understand from you and I really wanna see your face for god sake!!! God bless you
My guy, l appreciate how you break things down for easy comprehension
Since Freshman year of highschool I have been watching this guy, just helping me with algebra 2. Now I'm in AP chem and ap physics and ap cal as a junior and not dying, I have A's in all of those classes thanks to you. You sir are a legend.
omg you are the best! Thanks to your video I was finally able to learn how to solve the problem with enthalpy changes! :D
Such a great help! Thank you!
I believe Mr.JG is a gift of the Universe to us and all those who think they can cram and find out they can't then cry their eyes out and come here for salvation.
You gotta have an app for everything man youre amazing!
This teacher is a pro...thank you so much🙏
can i just take you with me to the exam ! YOURE GREAT GREAT GREAT THANK YOU !!!!!!!!!!
Thank you so much sir, this helped me FINALLY complete my assignment. Keep doing what you are doing. A literally saviour
Thank you so much bro...now im more understand in learning organic chemistry👍🏻👍🏻👍🏻
You motivated me...brother... Now i enjoy studying
Your classes are so brilliant! ❤
You make it more easy to understand than my chem tutor. Thank you
I only needed to much 5 minutes of this video to understand enthalpy 💌💌
*The man is a GENIUS*
Appreciate your videos, it was well explained. You're awesome! THANK YOU!
This has helped me beyond words!!!
Did u know, u make my whole world complete? I came in to college without chemistry, and I had to watch ur videos to understand before a tests or final exam and u, my friend, is a God sent ANGEL😇😇😇. I hope ur able to read this 😊😊😊
I dunno but I kinda love your voice u know the tone of it is really calm
thank you for the video
really appreciate it
listen my man. YOU R THE BEST!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
28:33 In this moment why you didn’t divided by 1000 ? How you converted a q from J to KJ ?!
Professor missed classes in preovious weeks due to his bussines trip, so , he came back, did all 6 lectures in one week, and put exam in next week, such a chad.
thank youuu
you have helped me a loot
I love you. im getting As on my ib chem tests just bc of you. Alhamdulilah
Hey, great videos like always! One suggestion for a future video that I personally believe is lacking in the general chemistry world and is VERY unique and I believe help students learn/visualize general chem concepts better would be creating a video relating general chem major concepts to real world examples, for example how pressure/temperature relates to higher altitudes, and how a hyperbaric oxygen chamber relates to pressure/gas relationship, or how salt melts ice relating to freezing point depression formula. I have yet to see a video on youtube that can link major general chem concepts to real world applications. Just a suggestion, let me know what you think!
You're right. I'll consider it some day.
Jason Tuli university and high school students have labs for that exact reason.
I like your suggestion!!!! It'll help make this subject more relatable to students
Fantastic it is very useful for jee Mains and Advance.
This guy is a legend all around the world
this teacher is very good!!!!!
You are the absolute best ❤️
You are life savior!!!!!
Thanks a lot.
Thank you Organic chemistry! My professor huhu,
I appreciate your great service
this video is very helpful thanks👍
27:00 When we figure out the mass, shouldn't we also include the 30g of NaOH as it is part of the solution?
i was thinking the same. maybe he made a mistake
No the dissolution reaction happens in the water and you are just looking for the change in energy for the water.
You deserve an award
I FINALLY GET IT. I NEVER THOUGHT I WOULD. (If you don't understand it, try re-watching the video a few days after)
This comment should be pinned!!
Many people misunderstood some concpets and asking questions tht JG sir already answered in the video, only if they rewatch it they wouldnt sit with the problem for forever almost 😭😭
bro ur the best thx for your courses
Dude!!You r aweosmeeeee!!!
Great help!!
This man is a legend
Life saver bro!!!
U are excellent special teacher l hope all good to u❤❤❤❤❤
I barely passed a test because I missed this part. If I have seen this earlier, I would have gotten an A on that test. :(
9:10 Carbon is 2C
i thought so lol
2 years after graduating highschool and ppl still like this comment. What a time
@@khaledragheb6503 كم جبت نسبه ؟ و دخلت اي جامعه ؟ 😂❤
@@3bdalra7man_32 A*, AUS
@@khaledragheb6503 ؟؟
Thanks helped me during my Chemistry CT :P
Awesome videos!! thanks
At 8:48, Carbon should have a coefficient of 2...
Great bro
Hi hoping I can learn this before my exam tomorrow wish me luck!
my God you are such a great teacher.I understood just from one example..
BLESS YOU SIR!!!
man you are absolutely amazing, I can't find words to thank you
Exams will be tomorrow but I'm speedrunning this sh*t
Your Videos are really helpful it saved my life in College. One question I have is Do you have videos for Calculus 2 and 3? And physics 2 and 3? Basically the series
I've always been taught that enthalpy change was=Energy in - Energy out.
But here Its is Energy of the products - Energy of the reactants.
Are there any difference in the formulas?
OKAY, but I love the sound of your voice!!! What kind of accent is this?
MURIKIN
Hi! Are Delta Hsolution and Delta Hreaction the same value in dissolution? Please help! I know qrxn and qsolution are opposite(signs). I'm confused as to if DeltaHsolution and DeltaHreaction are different.
U ar z best
This guy is from the future👐☺️
To add to that, most standardized exams are moving away from plugging in numbers, and moving towards a deeper understanding of the concept.
i love you but hate hess’s law!!! thank you for all your help
Thank you
Hey is this endothermic and what would be the type of endothermic question asked I understand exothermic. But this wasn't that clear
This helps for AP chem thanks man teacher Didnt show us this 🙏🏻
You can actually solve "The enthalpy of combustion for benzene..." in an easier way usinh Hrxn = q/n
Thank you Sir
i'd really like to know the name of the person who gave me so many light bulb moments!! Thanks so much you r amazing!!
good lectures
this is the reason why im topping my exams tysfmmm
I love u man😭
god bless your soul JG
These videos are for two things:
Learn faster than your teacher teaches you with 2x video speed
Fall asleep with 1x speed
thank you
Wonderful
Can you please also use the equations cause when you tell us the equation and then proceed into solving the question by analyzing the answer using units it gets very confusing
Bless you
I have a question. Around 38:25, when you wrote "12 mol/6540 kJ", why if 6540 kJ not negative? ∆H = -6540, so what am I missing? Thank you!
So did you know now ? 🤔
same question 😅 why it is not negative
very late but i’m assuming that it’s because grams can’t be negative? only thing i can think of.
Very easy to understand 👏
Hey could please explain if you understood at 36.19 that why is it -6540KJ and not -6540KJ/mol
At constant pressure reaction, the change in enthalpy is the heat transfered yes?