How to Find the Wavelength, Frequency, Energy and Photons | Study Chemistry With Us
Вставка
- Опубліковано 17 лют 2020
- I'll teach you how to calculate the wavelength, frequency, energy, number of photons, energy of mole of photons and more. We'll go over how to properly use each formula used in the electromagnetic spectrum and of course, everything is step by step.
📝 DOWNLOAD THE STUDY PLAN
melissa.help/studyplan
📗 FREE CHEMISTRY SURVIVAL GUIDE
melissa.help/freechemguide
📘 FREE ORGANIC CHEMISTRY SURVIVAL GUIDE
melissa.help/freeochemguide
💯 HERE'S HOW TO PASS ORGANIC CHEMISTRY 🎉
chemmunity.info/youtube
👉 MORE CHEMISTRY RESOURCES I CREATED 👈
melissamaribel.com/
🎓 CHECKOUT MY COMPLETE CHEMISTRY GUIDES:
📕 Thermochemistry Guide
melissa.help/thermonotes
📗 Acids and Bases Guide
melissa.help/acidbase1notes
📘 Naming Compounds and Acids Guide
melissa.help/namingnotes
📙 Dimensional Analysis, Significant Figures, and Density Guide
melissa.help/sigfignotes
📕 Gas Laws Guide
melissa.help/gaslawsnotes
📗 Stoichiometry Guide
melissa.help/stoichnotes
📘 Redox Reactions Guide
melissa.help/redoxnotes
📙 Molarity Guide
melissa.help/molaritynotes
📕 Limiting Reactants Guide
melissa.help/limreactnotes
📗 Lewis Structures Guide
melissa.help/lewisnotes
📘 Kinetics Guide
melissa.help/kineticsnotes
📙 Titrations Guide
melissa.help/titrations
📕 Matter, Atomic Structure, Empirical and Molecular Formulas Guide
melissa.help/matterguide
🙌 This was my go-to homework help when I was in school. Chegg Study is one of my favorites.
melissa.help/cheggstudy
📚 I made the mistake of buying all of my textbooks, I wish I had the option of renting them. Thankfully you do, with Chegg Textbook Rentals.
melissa.help/cheggbooks
💁♀️ HI I'M MELISSA MARIBEL
I help students pass Chemistry and Organic Chemistry. I used to struggle with this subject, so when I finally graduated with a bachelor's degree in Chemistry, I became a tutor so that you wouldn't have to struggle like I did. I know that with the right help, YOU CAN LEARN ANYTHING!
DISCLAIMER: Some links in the description are affiliate links, which means that if you buy from those links, I’ll receive a small commission. This helps support the channel and allows me to continue making videos like this. Thanks for the support!
Questions Covered in this Video:
1) Calculate the energy of X-rays with a wavelength of 15.0 nm.
2) What is the wavelength in meters of an FM radio wave with a frequency of 102.5 MHz?
3) If the wavelength of a photon is 552 nm. What is the energy of one mole of photons?
4) How many photons are contained in a flash of green light (525 nm) that contains 189 kJ of energy?








I need to start watching these videos LONG before i have to take the exam LOL
Watching this vid at 1.5x speed before a test. Boy i sure do love depression
im doing it at x2.0
playing in hardcore mode
I love this format. Makes learning so much more bearable.
📍😣😘🎵😣😘
Thank you for all showing us how to solve Photon energy, you always make this easy to understand. Thank you very much for your time to help students from College and High School! It was fun doing all these problems!
you have SAVED ME for my chem test thank you so much!
Ms. Maribel, if it wasn't for you teaching Melissa I am not sure this semester would have gone so smooth. Thank you so very much!
Struggling through this with another student makes this more relatable. really enjoy (I guess you can call it that) this format.
You are a great teacher!! God bless you , it's notorious your passion for teaching . You are an awesome pillar to learn from , thank you!!
wow you are such an incredible teacher, the way I'm learning this so quickly, thank you for doing what you do
my teacher literally showed these formulas; talked about electromagnetic waves and the spectrum; then never touched them again and now she's saying we should know it for our final :). Melissa, you're a literal angel
Thank you Melissa Maribel. You make it easy.
I loved it ! I just finished doing all the problems with you guys and to be honest I enjoyed it!
Yay!! I’m so glad you are loving this series 🎉 There are plenty more videos to come 😊
@@melissamaribel wwe
Thank you for this! I have a quiz tomorrow
Thank you, Melissa 💗
I wish I could hug you lol !! All your videos are extremely helpful, don't know what I would do without them!
This is super helpful!
I have an exam on Monday on everything you're covering in this series. Thank youuuuuuu for doing this.
Thanks for sharing these videos.
Love this!
I am starting all online chem April 13th and I’m so nervous because I am HORRIBLE at math and everything that has to do with it. I can relate to Melissa so much lol. Thank you for these videos! I am looking forward to watching your videos when I start my class. I also bought your Flashcards so hopefully I can pass my class 👍🏻
Then imagine doing this in the 10th grade without a calculator 😢
You are saving my chemistry grade! Thank you very much!
Thank you so much!
okay so I see I'm not the only one getting to the power of -33 when find wavelength of 15.00 nm. Does anyone know why this is?
thanks for sharing
😍
Thank you !!!
how are you getting the -17 exponent, i get the 1.3252 but I am getting -30 exponent on step 2 the first equation
My chem professor is amazing! She's giving us all the formulas we just need to know how to use them.
ok this actually makes sense now why do professors make it harder than it needs to be
can you explain how 10 e-34 and 10-e8 give you 10 e-17 for question number 1?
I think she made a mistake
did you get 1.33x10^-33 ?
I made a mnemonic to remember that c= lambda x frequency (v) formula
Cathine Likes Vulpix. where, V= frequency and L= lambda which is the wavelength
To all of them who are confused by that Vulpix thing. It's a pokemon. Gotta catch'em all!!
I like this one!!
I used to loathe math as a kid. That was before I began interest in discovering which synchronicities act as a direct map to the laws governing universal patterns of creation.
The electromagnetic spectrum is a great way to start looking into how to solve for other energetic equations pertaining to how the rest of the universe is structured.
It sounds far more complicated than it actually is. Maybe we can work together for making coloring books for this stuff some day. ,😊
exam soon this helped so much
Yaaaa this is very helpful well Mam can we get the pdf of this and before video Please Please Please Thank You So Much☺️
Super explanation 👍👍👍 well-done 😘😘😘 🇮🇳🇮🇳🇮🇳🇮🇳
How are people able to video chat with you? I feel this could really help someone with one to one question asking.
Is it possible to sum energy of the photons of green light with the energy of the photons of blue light algebraically?
Does anybody know the notes or past videos in which she talks about this?
How did you get 17 for the exponent
Do I have to change my name to Melissa before absorbing this information?
can anyone explain to me how to compute 1s^-1 on the calculator
This video is good, but you do better when you cover the topic rather than another's problem.
please can u help me with this question A ..Ozone formation in the upper atmosphere stars when oxygen molecules absorb UV radiation with wavelength less than or equal to 242nm. find the frequency and energy of the lest energetic of these photon . B... Ozone absorbs radiation with wavelength in the range 2200 A to 2900A ,thus protecting organisms from this radiation . find the frequency and the energy of the most energetic of these photons
is it possible to have a a digital calculator on the screen share. I add up all the numbers and still get a different answer or even though you lay it out, I still don't know how you got the answer. I don't know how to use my calculator. Can we see you plug it in?
Great idea! I’ll keep that in mind if I do a series like this again. I do have a video on some common scientific calculator mistakes if that helps at all ua-cam.com/video/ltww5INnliY/v-deo.html
does anyone know how she got 10^-19 at 25:35? im so confused on where that 19 came from..
:(
Do you know at the minute 16:47 she got 1.025x10^8 s^-1 and why i got 1.025x10^5 ???
Hello! I am sorry I am a bit late but I am assuming this may be a calculator issue? When I first started chemistry I would have typed that like (6.626) x (10^-34) etc... Make sure you aren't doing this! There is an exponent key on most scientific calculators (EXP) which is how you should enter the equation. Granted you are doing this I believe you should get 10^-19 as an exponent. Removing the calculator altogether you can look at it logistically.
I am assuming you know that you can subtract exponents in the numerator from those in the denominator, so, if I were to add the numerator exponents (-34 + 8) that would equal -26. I now can *subtract* that from the denominator's exponent, so (-26 - -7). The double subtraction symbols reduce to an addition symbol, and the ultimate exponent becomes (-26 + 7) = -19.
That is a bit difficult to convey in a text medium but I hope you understand! If you need any further help let me know! I wish you well! :)
@@michellecampa9850 I don't know if anyone answered you yet, but I figured it out, 102,500,000 represented in scientific notation is 1.025, to convert 102,500,000 to scientific notation we have to keep moving the decimal until there is only one non-zero digit to the left of the decimal point, in that case it would be 1, and if you count how many places we had to move it would be 8, so in this case its 1.025 and we moved 8 places, hope that helps!!
@@michellecampa9850 1.025 x 10^6 is the conversion which equals 102,500,000 or 1.025 x 10^8 because you move the decimal 8 places to the left to make 1.025 s-1
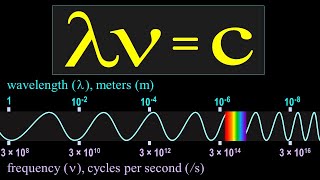
Speed of Light / Wavelength= Frequency
Frequency x Pi x 2=Angular Velocity of an Electron
Wavelength x 0.31831= Diameter / 2= Radius
Speed of Light / Radius = Angular Velocity
Radius x Angular Velocity = Speed of Light
Frequency / 241799050402293 = Electron Volts
Omg Melissa I wish I was lucky like her,I'm in a similar situation...sighs...😔😪great video tho
Play the frequency of the speed of light
At around 1:29, when talking about the first formula... you say that it helps to determine the frequency when given the wavelength and vice versa when given the wavelength and asked to find the frequency..... I assume you meant to say that when given the frequency we can find the wavelength? Should probably throw an edit on there for us confused folks trying to learn. Those are the same thing lol. 😂😂
I l❤ve u
Hmm I'm confused about the first example when I computed everything into the formula I got 1.33 x 10^-33 not 1.33 x 10^-17. Can you explain that part to me?
Me too!
@@isabellablanco959 Are you guys by any chance in 2nd year chemistry
i got 1.33 x 10 -17
did you ever get a reply to your question? I got 10^-33 too. im confused on the problem at 25:35 too. i couldn't get -19
@@89misselle YO do yll know why? I got 10^-33 too tf?
can you help me in this
Me pls. Can i have a help with😣😊💋
I am soo lost in chemistry this is depressing
Subtitle Indonesia 😭