Formal charge | Molecular and ionic compound structure and properties | AP Chemistry | Khan Academy
Вставка
- Опубліковано 16 гру 2019
- Keep going! Check out the next lesson and practice what you’re learning:
www.khanacademy.org/science/a...
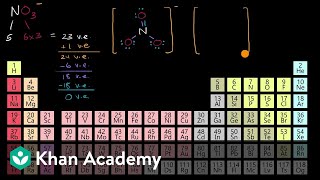
The formal charge of an atom in a molecule is the charge that would reside on the atom if all of the bonding electrons were shared equally. We can calculate an atom's formal charge using the equation FC = VE - [LPE - ½(BE)], where VE = the number of valence electrons on the free atom, LPE = the number of lone pair electrons on the atom in the molecule, and BE = the number of bonding (shared) electrons around the atom in the molecule. View more lessons or practice this subject at www.khanacademy.org/science/a...
Khan Academy is a nonprofit organization with the mission of providing a free, world-class education for anyone, anywhere. We offer quizzes, questions, instructional videos, and articles on a range of academic subjects, including math, biology, chemistry, physics, history, economics, finance, grammar, preschool learning, and more. We provide teachers with tools and data so they can help their students develop the skills, habits, and mindsets for success in school and beyond. Khan Academy has been translated into dozens of languages, and 15 million people around the globe learn on Khan Academy every month. As a 501(c)(3) nonprofit organization, we would love your help!
Donate or volunteer today! Donate here: www.khanacademy.org/donate?ut...
Volunteer here: www.khanacademy.org/contribut...








You are one of the only mathematicians I understand on UA-cam. You have truly helped me many times. You have my subscription
lier
Million thanks for making science & maths easy for me and many others.
Regards.
thank you so much for the help you give everyone, truly appreciate it!
Mesmerizing classes with colorful lessons.
Jazakallahu Khairan.
Really great
Tecnology just like education revolution these days
Hatsoff 🙌
I always find it difficult to memorize this formula, thanks for making it so easy!
made it laughably easy as compared to the McGraw hill textbooks im paying hundreds of dollars for every semester. Thank you loads
This is a good video explaining the concept of formal charges. Are there more such examples around?
This is amazing.
Good Job!!!
very helpful explanation
yaaay gooo khan!!!
Even though they have a different formal charge they're still the same chemical right? If we were to some this up to a complete mathematical equation they would both come out to zero. I guess the point I'm trying to make is what use do we get out of a set up like this?
Saved me!
Great
Only I have to say that
Biiiiig thanks
In the first structure, how does the last oxygen have a formal charge of-1? Because it can’t have 7 electrons in the outer shell and the octet rule is not satisfied
Actually, there are elements which are stable despite not obeying the octet rule. That's why the rule is discarded in higher classes, so we study about hybridisation.
It does have 8 electrons in its valence shell (6 lone pair electrons + 1 single bond (2 electrons))so its octet rule is satisfied however it has only 7 of those electrons allocated to it due to the fact that you take 1/2 of bonded electrons so its 6 lone pair electrons + 1 electron from the single pair so its 7.
Third.
Second
can anyone help me in this problem ?
A driver at 25 m / s detects an obstacle in his lane. Braking starts at a distance of 83 m. At a distance of 23 m, a speed of 13 m / s remains. The speed is expected to decrease steadily.
a) What is the absolute value of the acceleration of the car? Give the answer with three significant digits.
You need to use the formula v^2-u^2=2as here, v is 13m/s and u is 25m/s. 'S' is the distance and 'a' is acceleration. Acceleration in this case will have a negative value (retardation)
Wait, I just realised that this comment was posted 3yrs ago😅
First
Thanks so much for this, it seems like magic to me in class 🥲. Bless up chef