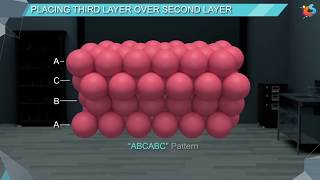
Packing fraction of hexagonal close packing system in three dimension(3D)-Solid State chemistry
Вставка
- Опубліковано 10 лис 2020
- #solidstatechemistry
#packingfractionofhcp
#hcp3D
#effectivenumberofatomshcp
#hexagonalclosepacking
#Volumeofhcp
#V=24root2r3
#JEEchemistry
#NEETchemistry
#NCERTChemistry








Thank you mam, you cleared my whole confusion about this topic
Amaizing video !
Amaizing video✌👍
v good to listen and learn
great work i couldn't learn it in my classes but you explained it so well that i can make someone understand it
Great work!
Amazing explanation..
Gem of an explanation, ma'am! thanks.
thank you so much, mam.a picturesque explanation indeed.
Thank you so much mam❤❤..
Thank you so much for your detailed explanation!!! You made it easy to understand.
very good!
Super
Excellent explanation ma'am 🙏
Good information mem
Men Plz Help Mi WhatsApp No
Nice ❤
💯💯💯
Excellent mam🎉
Thanking You very much Mam.
i love u
u saved my midterm
BEAUTIFULLU EXPLAINED
Thank you very much!
God bless you
Thank you very much
Thank you mam🥰😍
madam u r my hero
Nice explanation join pw mam ❤️❤️
Very great and accurate mam ( actually I got many video but they were doing wrong even though one person answer as 91%😂)
bro it was of 2d hcp unit cell
@@IMPULSE17 no no , she is also going the same 3d only with the same structure
Fiinal answer is using π/3√2= 0.604 so paking factor is 60.4 ... But in hcp is 74 ...
i think she made a mistake on area of hexagon should be 3 times root3 divide by 2 times r^2
You are better than my Professor.
why is the triangl´s area √3 r^2 and not √3 r^2 /4
here, r is the radius of the atom, not the length of an arm of the triangle. And length of one arm of triangle is double the radius of atom. So, if you substitute the value of side of a triangle with radius of the atom, the 4 in the bottom got vanished.